Unveil the true standard of your cultivation and expand your reach with TRUSTT's GACP Audit
In a market where claims often outpace reality, TRUSTT is your partner in not just asserting, but proving and enhancing your compliance with EU-GACP standard, allowing you to unlock Eurpean markets and beyond.
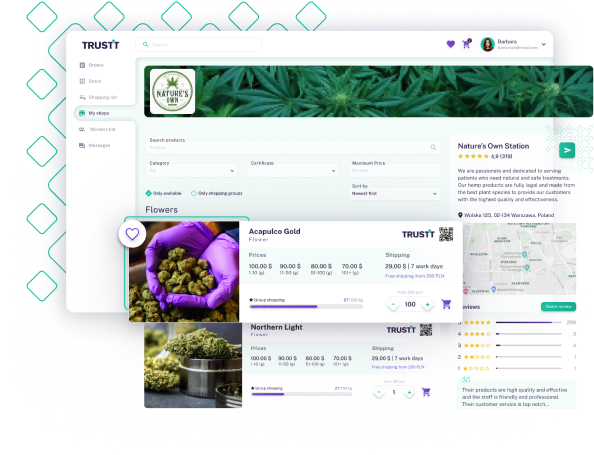
Don't declare it, prove it.
Let’s face it – the declaration of GACP compliance is widespread, but how often are these claims validated?
Infrequent audits with a broad brush can overlook the nuances of batch-level compliance, leaving both cultivators and their business partners in the dark. At TRUSTT, we illuminate these details, bringing transparency and continuous compliance to the forefront of your operations.
Our GACP Audit is meticulously designed to delve into your processes, ensuring that every batch meets the highest criteria for safety and quality, safeguarding consumer trust and your business reputation.
Solution that will help you sell more while increasing your profitability.
Our GACP audit isn’t a one-time checkmark; it’s a comprehensive dive into your cultivation process, resulting in a detailed report that outlines non-conformities and guides you towards rectification and excellence.
And it doesn’t end there – with TRUSTT, you receive a certified Corrective and Preventive Action (CAPA) plan, and an opportunity to manage ongoing compliance through our GACP Compliant TRUSTT Seed to Sale Software.
- Operational efficiencies resulting in cost savings and higher profitability
- New markets and customers by demonstrating GACP compliance
- Better crop management and increase in yields quality and size
- Paperless track & trace compliance
- Premium Pricing Opportunities
- Enhanced Brand Reputation and customer confidence
- Competitive Advantage
- Easier regulatory compliance

Schedule Your FREE GACP Audit Consultation


Audit Outomes
- Detailed audit report with non-conformity list.
- TRUSTT certified CAPA plan
- Discounted Subscription to our GACP compliant Seed to Sale solution for ongoing standard monitoring.
- Premium Access to a Trading platform for data-verified producers
Post-audit, choose your path – implement our certified CAPA plan independently, or let our Seed to Sale and Cultivation Module Solution become your digital companion, ensuring real-time access and continuous verification for each batch.
This is compliance made convenient, keeping you a step ahead in an ever-evolving industry.
Basis of TRUSTT GACP Certification
The scope of TRUSTT GACP covers medical Cannabis propagation, cultivation and post-harvest processes and fully complies with both the WHO and EMA GACP guidelines.
- The World Health Organisation (WHO) guidelines: WHO Guidelines on good agricultural & collection practices (GACP) for medicinal plants – Geneva 2003
- European Medicines Agency (EMA GACP): guideline on Good Agricultural and Collection Practice (GACP) for starting materials of herbal origin (July 2005)
- EU GMP guidelines by European Medicines Agency (EMA): Regulation No. 1252/2014 and Directive 2003/94/EC, applying to active substances and medicines for human use
Certified organizations will receive both TRUSTT GACP certification and GACP compliance certificate for both the WHO as well as EMA GACP guidelines.

Step by Step to your GACP...

Why our Cultivation Module?

Grow Consistency
Cannabis complexity, (over 500 active substances) and high sensitivity to environmental factors impact chemical composition of a final product.
Quality Control
Over 600 Standard Operating Procedures related to cultivation with number of other quality and legal documentation that must be available at any time.
Regulatory Compliance
Following EU-GxP standards is challenging, especially taking in account jurisdiction variations.
Staff Empowering
Processes related to cultivation quality and compliance are difficult to follow and upkeep by staff leading to errors.
Bring your Cultivation to the next level!
Book free consultancy call now!


